First off… what is Real-World Evidence (RWE)?
Real-world evidence is the clinical evidence regarding the usage and potential benefits or risks of a medical product derived from analysis of real-world data (RWD). RWE can be generated by different study designs or analyses, including but not limited to, randomized trials, including large simple trials, pragmatic trials, and observational studies (prospective and/or retrospective).1
Why is RWE so important?
RWE provides a more comprehensive understanding of how a therapy works in the “real world” outside of randomized controlled trials (RCTs). For example, researchers and HCPs can see how efficacious treatments are for different patient sub-groups that weren’t included in RCTs – ultimately enabling them to make more customized treatment plans for patients.
Case Study: See how one pharma company leveraged real-world evidence in their physician engagement program
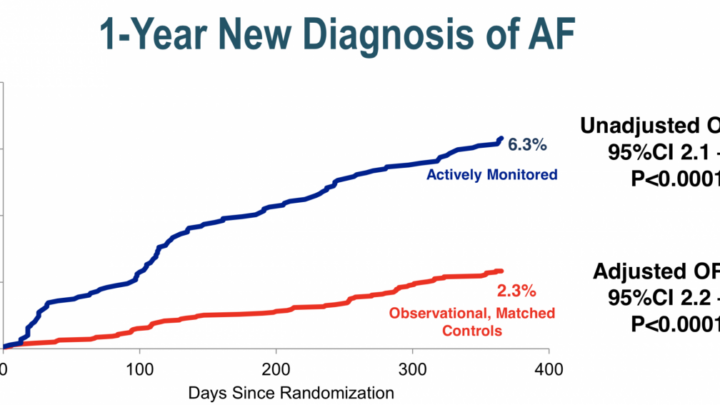
Campaign Objective: Educate physicians about the results of a real-world evidence based meta-study of 430K cardiology patients
Sermo Solution: Interactive Post leveraging unbranded video and poll questions
Target: Italian Cardiologists and PCPs
Campaign success metrics
- 2x more video views than goal
- 50% more views than goal
- 15 percentage-point increase over engagement rate goal
Ad impact & effectiveness study results
Measurement Objective: Understand ad awareness and message recall of educational ad
Methodology: 5-minute online survey to test and control groups, fielded June 2021
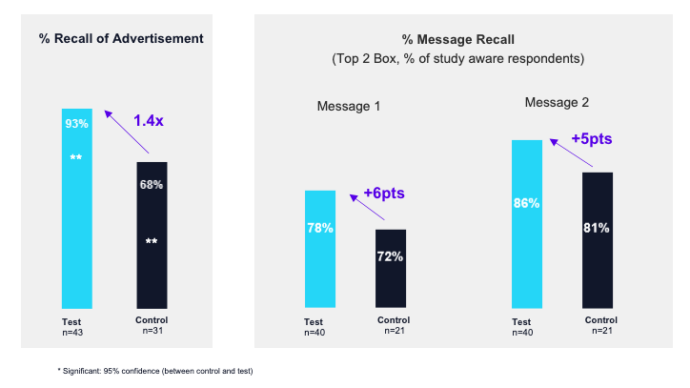
The results?
The Sermo RWE campaign had near universal recall and effectively increased message recall among targeted physicians, signaling this is an extremely effective content strategy for educating HCPs and supporting their treatment decision-making.
To learn more, contact us at business@sermo.com
1Source: https://www.fda.gov/science-research/science-and-research-special-topics/real-world-evidence
Hello, we’re Sermo! We appreciate you checking out our blog post Leveraging real-world evidence to drive physician engagement. This exclusive content is built to educate our readers on the importance of real-world evidence to drive physician engagement.
We turn physician experience, expertise, and observations into actionable insights for the global healthcare community. Engaging with more than 1 million HCPs across 150 countries, we provide physicians with a social platform that fosters impactful peer-to-peer collaboration & discussions about issues that are important to them and their patients. Sermo offers on demand access to physicians via a suite of propietary technology to provide business intelligence that benefits pharmaceutical, healthcare partners and the medical community at large.
Interested in learning more? Check back any time and follow us on Facebook, Twitter and LinkedIn for the latest and greatest in healthcare insights.
To explore our exclusive business solutions, please visit us at sermo.com/business or email us business@sermo.com